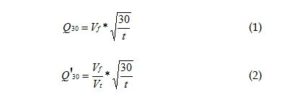“Baroid” Test Procedure
Introduction
The Baroid test is intended to determine the filterability of a raw sludge and, if necessary, inorganic chemical conditioning. The most common inorganic chemical conditioning is ferric chloride and lime. The Baroid is a relative test in that can determine which dosages result in improved filterability but it can not be used for filter press sizing.
Description of Equipment and Outline of Baroid Test
The Baroid is made up of a stand and a three piece bomb unit. The stand has a threaded screw that is used to tightly close the bomb unit so that compressed air or sample does not leak out. The bottom piece of the bomb has an outlet in the bottom center with a screen to support the filter paper. A flat rubber ring is used to provide a seal between the bottom and center section of the apparatus. The top piece has another rubber ring seated in it and has the air inlet port.
A filter paper with the rubber ring on top is place on the screen in the bottom piece and the center section is placed firmly on top of the paper. The sludge (raw or conditioned) is poured into the Baroid and the top is put on and the unit is placed in the stand and screw is tightened. Compressed air, at a constant pressure (usually 100 psig), applied through the top inlet. Filtrate is collected until either 7.50 minutes have elapsed of the air comes out of the bottom outlet, which ever occurs first.
After the test is done the filtrate volume is measured, the filtrate quality is determined, the cake quality is determined, and the cake release from the paper is determined. The last three parameters are estimated visually on a 1 – 5 scale per descriptions given later.
Chemicals
Ferric chloride solution of known concentration and density
Hydrated lime, chemical grade with analysis of 90 – 95 % as Ca(OH)2 or 68 – 72 % available CaO
The hydrated lime should be made up as a 10% w/w slurry to add to the sludge. Normally lime is added to sludge as a 10 – 15% w/w slurry.
Other inorganic chemicals such as alum as specified by the customer.
Detailed Procedure
- Obtain a 5 to 10 gal representative sample of the sludge that will be used for the filter press testing.
- Determine or obtain the feed solids and pH of the sludge. If a laboratory sample is needed collect a 1 liter sample for analysis.
- Using a 250 ml sludge sample add the chemical conditioning per Table 1
Table 1. Recommended Chemical Dosages for Each Baroid Run
| Run | Ferric Chloride Dose % | Lime Dose As CaO % |
| 1 | 0 | 0 |
| 2 | 5 | 20 |
| 3 | 5 | 25 |
| 4 | 5 | 30 |
| 5 | 6.5 | 20 |
| 6 | 6.5 | 25 |
| 7 | 6.5 | 30 |
| 8 | 8 | 20 |
| 9 | 8 | 25 |
| 10 | 8 | 30 |
| 11 | 10 | 20 |
| 12 | 10 | 25 |
| 13 | 10 | 30 |
| 14 | 10 | 35 |
Note that chemical dosages are conventionally reported for ferric chloride and lime on a w/w%, dry solids basis.
- Add ferric chloride to the sludge and mix thoroughly
- Add the lime slurry to the sludge and mix thoroughly
- Pour into the Baroid bomb and put top on the bomb
- Put the bomb in the stand and tighten the screw.
- Put a beaker underneath the filtrate outlet to collect the filtrate
- Attach the compressed air source to the inlet
- Start timing the run when the air valve is opened
- Stop test when either 7.5 minutes have elapsed or air has is coming the filtrate outlet, whichever occurs first
- Record the volume and elapsed time and note the filtrate quality (see Table 2)
- Disassemble the unit and inspect the cake and cake release (see Table 2)
Table 2. Description of Ratings for Filtrate, Cake, and Cake Release
| Rating | Filtrate Quality | Cake Quality | Cake Release |
| 1 | Looks like unfiltered sludge | No cake formed or unfiltered sludge on top of cake | No cake |
| 2 | Very dirty | Cake is very soft, can squeeze water out of cake by hand | Cake sticks to the paper and must scrapped off the paper |
| 3 | Dirty | Bottom cake is firm but most of the cake is soft | Cake releases from about half of the paper |
| 4 | Small amount of visible solids | Cake is mostly firm, top is tacky | Cake releases from almost all the paper |
| 5 | Clear, no visible solids | Cake is firm throughout | Cake releases from the paper |
- Repeat steps 3 through 13 varying the chemical conditioning dosages.
- If the recommended dosages do not give a satisfactory cake and filtrate, vary the chemical conditioner dosages based on the direction of the best cake and filtrate observed repeat steps 3 through 13 with the new dosages.
- If there is information indicating that different dosage ranges should be examined test these ranges then, if necessary, test either the recommended ranges of other ranges as outlined in step 15.
Calculations
The basic calculations are
where
Vf is filtrate volume in ml
Vt is total volume of conditioned sludge in ml
And t is time in minutes.
Equation 1 was developed for evaluating drilling muds and assumes a constant total volume. Equation 2 corrects for the variable volumes common with chemical conditioning and uses the volume ratio. Thus the total filtrate volume changes do not mislead one when the volume ratio is essentially unchanged after considering the total volume of conditioned sludge.
Interpretation
As the chemical conditioning improves the filterability of the sludge both the Q30 and Q‘30 will increase. As these values increase a break point should emerge where increasing the chemical dosages does not significantly improve the filterability. Once this “break point” is reached the cake release, cake quality, and filtrate quality parameters will normally be 4 or 5 and will not change significantly once the “break point” is reached.
Very thick cakes are prone to have tacky tops and very hard bottom layers. This may mislead an inexperienced analyst to conclude that more conditioning is needed when in fact the sludge will dewater very well in filter press. The thinner the cake the more consistent the cake tends to be throughout.
There is no one value that indicates an optimum chemical conditioning for all sludges. Each sludge has its own value for optimum chemical conditioning. These values depend on the type of sludge, sludge concentration, and sludge age. The Baroid test is qualitative in nature and semi-quantitative for a specific sludge at a specific site.